 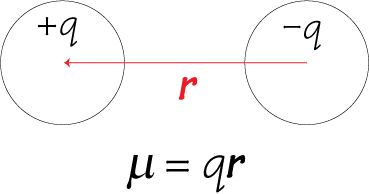
The end that is mawr less electro negative. The end that is more electoral negative gets a partial charge of Delta Negative. If you were to calculate the difference between flooring and carbon, you would see the difference is greater than 0.4. This is illustrated by a die poll arrow that always points towards the Mawr electro negative element. Now, if you have polarity in a bond, you have a dipole moment. Difference in electro negativity, which is Delta e n equals the higher electro negativity value minus the lower white. Not polarity is just the unequal sharing of electrons between these bonding atoms at a difference in electro negativity, greater than four is considered significant now. Now, a dipole moment is the polarity that arises when elements in a bond have a significant difference in their electoral negativity. So click on the next video and let's start talking about dipole moments 
So just remember as we start talking about dipole moments, you need to remember that it's related to the electro negative values of these different elements. And realize that over here in the bottom left corner we have francie um which has the lowest electro negative value. So because they're so unstable, we don't give them electro negative values. Krypton being three point oh and Xenon being 2.6 here, most of the seventh row is comprised of synthetically formed metals. So we tend not to talk about their electro negativity ease, but because Krypton and xenon are lowered down and have larger shells involved, they sometimes can have electro negative values associated with themselves. Now recall that noble gasses are perfect. So if we take a look here in terms of this periodic table, we'd say that we can see that the non metals, those in blue have a higher electro negative value, with flooring being the most electro negative. And the periodic trend that you need to remember is that electro negativity increases, moving from left to right across a period or row and up a group. Is a measurement of an element's ability to attract electrons to itself. Now recall electro negativity, abbreviated as E N. We first need to revisit electro negativity. To understand the dipole moment found within either a chemical bond or chemical compound. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
